|
This property of water is critical for all life on earth. That means that about 10 percent of an ice cube (or iceberg) will be above the water line. In things air density, this matters because it is one of the steps towards scientists assessing the chemicals in that air, including toxins and imbalances, like too much carbon dioxide. The density of ice is about 90 percent that of water, but that can vary because ice can contain air, too. In other words, how much something weighs multiplied by how much space it occupies. air (in its gaseous form) has a density of 0.0012 g/cm³ The solid (steel) has the highest density, the gas (air) has the lowest density, and the density of the liquid (water) is in. Present Formulation In the following, the air density equation derived by Jones 2 will be incorporated into a refractivity equation. Equations (5), (6), and (8) are generally combined in the calculation of the refractivity of moist air, in the visible region. Pressure varies from day to day at the Earth. With fewer air molecules above, there is less pressure from the weight of the air above. High in the atmosphere, air pressure decreases. The mass of 10 m 3 air can be calculated as. 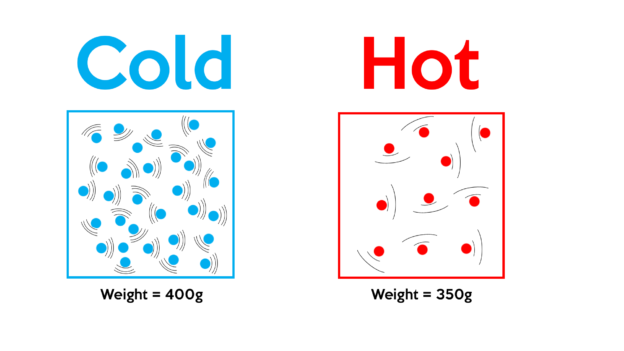
From the table above - the density of air is 1.205 kg/m 3 at 20 o C. That means air exerts 14.7 pounds per square inch (psi) of pressure at Earth’s surface. density (kg/m 3) Example - Mass of Air at Temperature 20 o C. Densely populated cities often have poor air quality because of the air. Above every square inch on the surface of the Earth is 14.7 pounds of air. Air quality therefore measures the density of air pollutants, such as smoke and emissions. How do you calculate density in standard conditions 1. Air quality is defined by the number of pollutants in a certain area. The value of air density is 1.29 grams per litre (0.07967 pounds per cubic foot) at 32 degrees Fahrenheit (0 degrees Celsius) at average sea-level barometric pressure (29.92 inches of mercury or 760 millimetres). This can apply to gases, liquids, and solids. If the humidity is high, the air is less dense - does this mean a golf ball would travel farther in higher humidity (given the same amount force)? Or would the water molecules slow the ball down Density is, in really basic terms, mass times volume. for density ('rho') generally in kg/m 3, mass m in kg and volume V in m 3. ( 720.775) is the density factor for standard air. Density can refer to the number of molecules in a substance. I can understand that as the humidity rises, there is less room for nitrogen and oxygen molecules in a given area and therefore the density is lower.Ī question my friends and I have though, pertains to our golfing. If it is more humid, with more water molecules in the air, is there more drag on a golf ball or less? 
Well I'm still wondering about the golfer's question.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
